Targeting Cancer Drugs More Effectively: First EU Project Integrates Pharmacogenomics into Tumor Boards
Joint Press Release from Bosch Health Campus and NCT Heidelberg
The Dr. Margarete Fischer-Bosch Institute for Clinical Pharmacology (IKP) at the Bosch Health Campus in Stuttgart is leading Europe’s first EU project to systematically integrate pharmacogenomics into molecular tumor boards (MTB). The goal of PGxMTB is to systematically incorporate patients’ genetic factors into treatment decisions, thereby making cancer therapies safer and more effective. The National Center for Tumor Diseases (NCT) Heidelberg is contributing extensive expertise and data to the project.
Stuttgart/Heidelberg, March 17, 2026 – In current cancer therapy, many drugs are specifically tailored to tumor alterations. However, even with well-matched therapies, severe side effects often occur or drugs do not work as expected—in part because genetic differences among patients have not yet been systematically taken into account, or because the tumor itself has characteristics that can play an important role in drug side effects during cancer therapy.
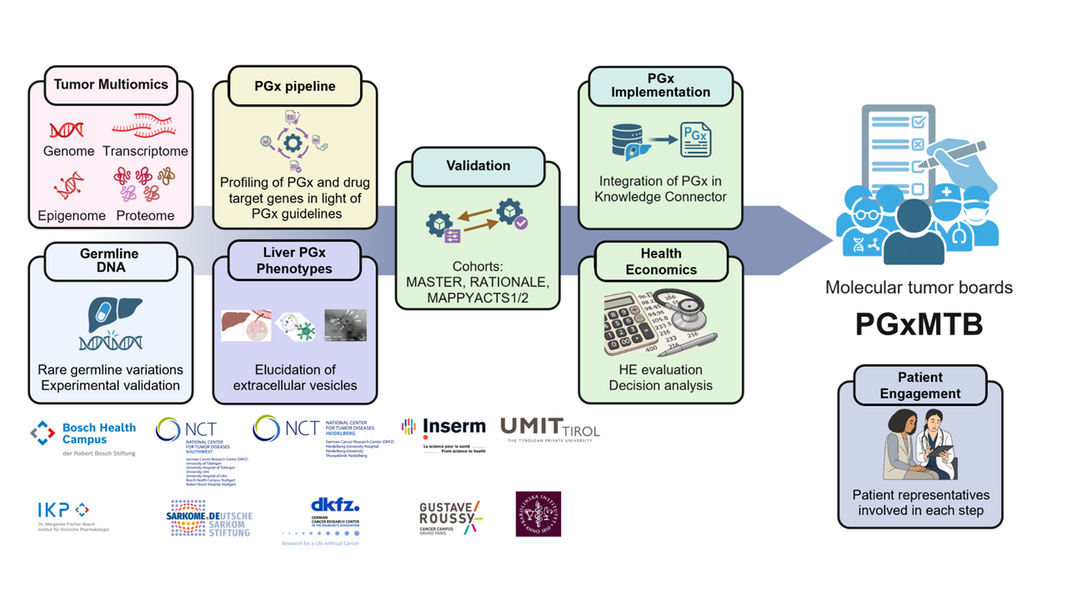
The international PGxMTB project, led by the Dr. Margarete Fischer-Bosch Institute for Clinical Pharmacology (IKP), aims to close precisely this gap. The IKP is part of the Bosch Health Campus, one of the supporting institutions of the National Center for Tumor Diseases (NCT) Southwest. The German project partner is the NCT Heidelberg, which contributes its comprehensive clinical expertise, diagnostic experience, bioinformatics development capabilities, and an extensive database to the project. The international consortium comprises a total of five scientific teams—in addition to the two German partners, the Gustave Roussy Institute in Villejuif, France; the Karolinska Institute in Stockholm, Sweden; and the Institute for Public Health, Health Services Research, and Health Technology Assessment in Hall in Tirol, Austria.
A unique feature of PGxMTB is active patient involvement facilitated by the German Sarcoma Foundation, which is an additional partner in the consortium. Together, they aim to systematically incorporate pharmacogenomic data into molecular tumor boards—interdisciplinary medical case conferences where complex cancer cases are discussed collaboratively—in the future. Over 300 genes relevant to the efficacy and tolerability of cancer drugs and supportive therapies are analyzed. For the first time, the individual genetic makeup of the patient is taken into account alongside the genomes of the various cancer entities.
“With the coordinated integration of pharmacogenomic data into clinical practice, we are breaking new ground—to date, no similar initiative exists in Europe that provides for this. This is a significant step toward further optimizing cancer treatment, reducing severe side effects, and making treatments more effective. By actively involving patients as research partners, we can make optimal use of their experiential knowledge,” says Prof. Dr. Matthias Schwab, project coordinator and head of the IKP at the Bosch Health Campus research division, as well as managing director of the NCT SüdWest.
At NCT Heidelberg, Prof. Dr. Stefan Fröhling, Executive Director at NCT Heidelberg and Head of the Department of Translational Medical Oncology at the German Cancer Research Center (DKFZ), and Dr. Dr. Daniel Hübschmann, Head of the Computational Oncology Group at NCT Heidelberg and the Innovation and Service Unit for Bioinformatics and Precision Medicine at the DKFZ, are responsible for PGxMTB. Among other things, the project utilizes existing data from large precision oncology initiatives such as the DKFZ/NCT/DKTK MASTER program. Based on this data, the consortium is developing and expanding a bioinformatics workflow. “We want to improve the treatment of cancer patients by working with our partners to tap into a biological level that has been largely ignored until now but is extremely important. Our goal is individualized and risk-adapted pharmacotherapy,” says Daniel Hübschmann.
Although pharmacogenomic tests are already established in certain indications, they have not yet been incorporated into tumor boards. PGxMTB is thus setting new standards by simultaneously taking into account both the patient’s genome and the tumor’s, and plans to implement IT-based tools, for example, in the molecular tumor boards in Heidelberg and at the Gustave Roussy Institute in Villejuif.
About the Bosch Health Campus
The Bosch Health Campus brings together all of the Robert Bosch Foundation’s institutions and funding activities in the field of health under the four pillars of Treatment, Research, Education, and Funding. With its interdisciplinary network of facilities and more than 3,600 employees, the Bosch Health Campus has made it its mission to offer innovative solutions to the major challenges facing the healthcare system.
The Bosch Health Campus includes the Robert Bosch Hospital, the Dr. Margarete Fischer-Bosch Institute for Clinical Pharmacology, the Robert Bosch Center for Tumor Diseases, the Robert Bosch Center for Integrative Medicine and Health, the Bosch Digital Innovation Hub, the Institute for the History of Medicine, the Irmgard Bosch Education Center, and the Robert Bosch Center for Innovations in Healthcare.
Learn more at www.bosch-health.campus.com
About the National Center for Tumor Diseases (NCT) Heidelberg
The National Center for Tumor Diseases (NCT) Heidelberg is a joint institution of the German Cancer Research Center (DKFZ), the University Hospital Heidelberg (UKHD), the Heidelberg Medical Faculty of the Heidelberg University and the Thoraxklinik Heidelberg. The aim of the NCT Heidelberg is to transfer promising approaches from cancer research into clinical practice as quickly as possible, and thus to benefit patients. This applies to both diagnosis and treatment, in aftercare or prevention. Participation in clinical studies opens up access to innovative therapies. The NCT Heidelberg is thus a leader in transferring new research results from the laboratory to the clinic The NCT Heidelberg, founded in 2004, is part of the NCT with further locations in Berlin, Dresden, SouthWest (Tübingen-Stuttgart/Ulm), WERA (Würzburg, Erlangen, Regensburg, Augsburg) and West (Essen/Cologne).
More at www.nct-heidelberg.de
Press contact:
Dr. Martin Staiger
National Center for Tumor Diseases (NCT) Heidelberg
Communications and Events
Im Neuenheimer Feld 460
69120 Heidelberg, Germany
Tel.: +49 6221 56-311272
Email: martin.staiger(at)nct-heidelberg.de
www.nct-heidelberg.de







